Since the orbitals are only telling us about the probability of finding an electron, it’s imperative to be as sure as possible about the whereabouts of the electron inside an atom. The Heisenberg Uncertainty Principle says – “ it is impossible to define with absolute precision, at the same time, both the position and the momentum of an electron” which rules out any definite orbit. To be able to assign a definite path, it’s absolutely necessary to know the location of the electron at an instant and to be able to predict its movement the next instant. Although from a simplistic view, electrons may look like going around the nucleus in a similar way, in practice, they populate regions of space which are called orbitals. Orbitals and orbits – the differenceĪ planet moving around the sun in a fixed path is said to be moving in an orbit. 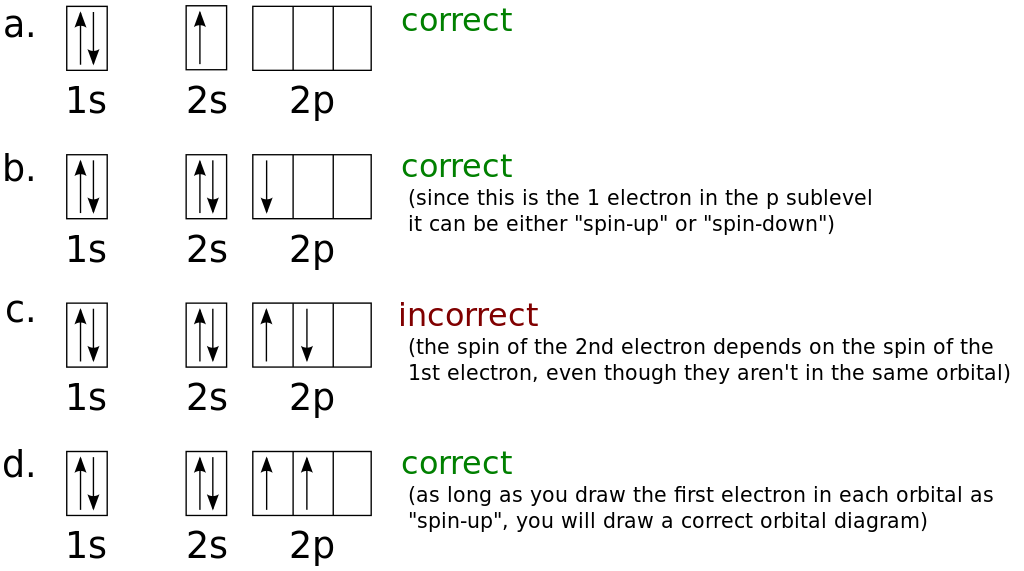
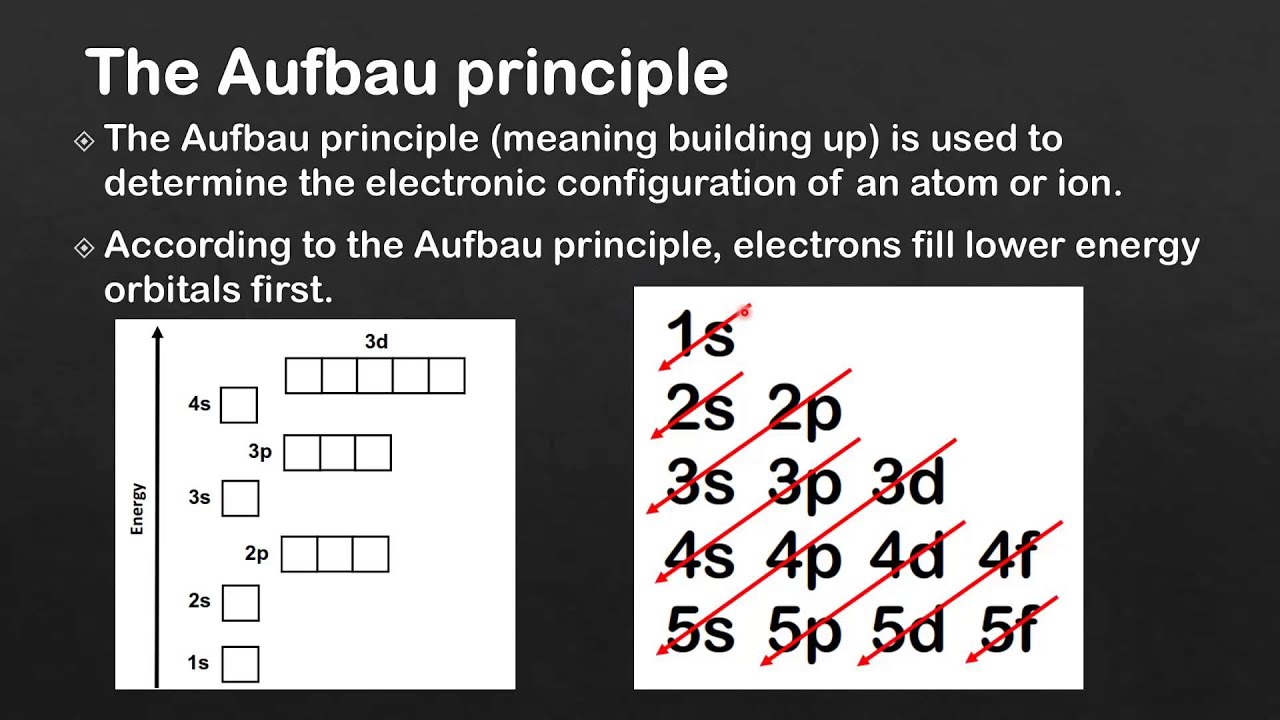
In order to understand the nature of electronic configuration, we need to understand the terms orbitals and energy levels. The very first criteria to explain the chemistry of any species, is to identify the arrangement of electrons because, the chemical reactions are essentially the rearrangement of electrons. Further development of chemistry, especially in the sphere of quantum mechanics, has enabled us to rationalise the existence of all elements in modern Periodic Table and their arrangements are applied to elucidate the chemical properties. In this endeavour, Mendeleev put forward the periodic table of elements. Since the very early days, chemists have tried to organise the elements so that their properties can be studied with minimum of effort. Which quantum number defines the energy of orbitals?.What do you understand by the term “Orbital?”. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
